- info@clinicalresearchofontario.com
- +1 (416)-648-6167
- Fax: (416) 332-8081
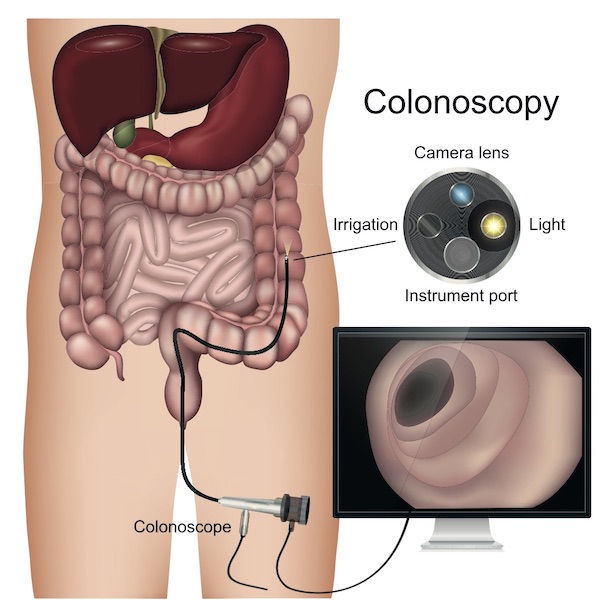
Colonoscopy is a direct visualization technique which allows both visualization and instrumentation of the entire colon under optimized conditions. It was first recommended in 1997 as a screening test for individuals reaching their 50th year. It is unique amongst screening modalities in its ability to detect, biopsy, and contemporaneously treat suspected pre-malignant and malignant lesions. Its use as a screening tool is recommended by all national gastroenterology and cancer societies of note in Europe and North America7. The efficacy of colonoscopy was established in a large multicenter study, the National Polyp Study, which showed a 76-90% reduction in CRC incidence 5.
Patient compensation is available for time.
Clinical research are research studies that are performed in patients to evaluate medical treatment and procedures. The goal of clinical research is to improve the diagnosis and treatments of patients and to improve the health of individuals who participate in the trials. Clinical trials help determine the safety and efficacy of medications, vaccines, diagnostic products and medical devices to all types of disease